Know in one minute about Porphyrins
|
Introduction
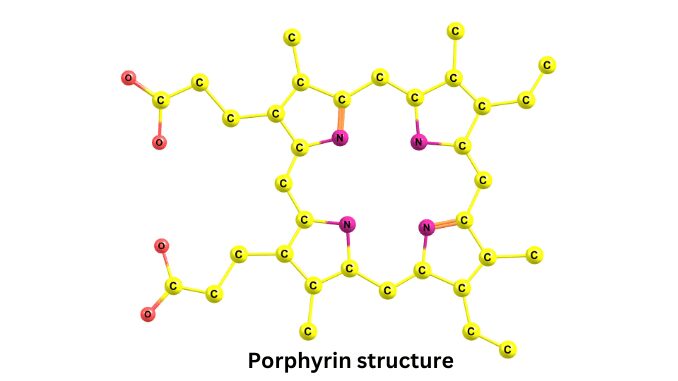
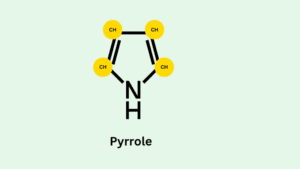
Porphyrins are complex structures. It is a large ring molecule consisting of four pyrrole subunits. Pyrrole is a smaller ring made from four carbon and one nitrogen molecule. Pyrrole subunits are interconnected from their alpha carbon atom by (=CH–) methyl bridges.
- Methyl bridges connect the pyrroles. These are connected through a series of single and double bonds which forms the molecule into a large ring.
- Four Pyrroles are connected is technically called tetrapyrrole. The ring is flat in space.
- The distribution of electrons around the ring is equal. For this reason, porphyrin is considered an aromatic compound.
- This means the molecule is very stable. Aromatic rings form the base of all porphyrin molecules.
Metalloporphyrins are a type of porphyrins. In metalloporphyrins, metal ions can bind with nitrogen atoms of pyrrole rings to form complexes. Where metals have not bound these types of porphyrins are called free porphyrins.
- Metal complexes derived from porphyrin occur naturally. Heme belongs to the metal complexes. It is a pigment in red blood cells (RBC) and a cofactor of the protein hemoglobin. Hemoglobin carries oxygen from all organs and tissues.
- Haemoglobin, myoglobin, cytochromes, and chlorophyll are the major example of metalloporphyrins. It is an iron-containing porphyrin while chlorophyll is a magnesium-containing porphyrin.
Properties of Porphyrins
- The cyclic compound is made with four pyrrole rings. Methylene bridges connect the pyrrole rings.
- Acetyl group (A) and propanol (P) group are present and attached to each pyrrole ring on the porphyrin molecules.
- Porphyrins vary from his side chains. The nature and distribution of side chains determine the physical properties of Porphyrins like solubility.
- They are highly stable compounds. They chelate with many different metals to produce metalloporphyrins. For example hemoglobin (chelate with iron) and chlorophyll (chelate with magnesium)
- Porphyrins can exist at varying Oxidation levels.
- Porphyrins emit intense red fluorescence when exposed to light at around 400nm. Thus spectrofluorimetric methods provide very sensitive detection and quantification of Porphyrins.
Classification of porphyrins and their functions
Based on the arrangements of the A (Acetyl group) and P (propanol group) groups the molecules are classified into two groups.
1. Type I porphyrins
Their substituent groups on the 8 positions are symmetrically arranged; they are known as type I porphyrins, e.g. uroporphyrin I.
2. Type III porphyrins
In there, substituent groups are asymmetrically arranged and are more common in the biological system. Type III porphyrins are found more commonly than type I.
The most important porphyrins in human beings are
Porphyrins |
Occurrence |
| Uro-porphyrins | Found in urine |
| Copro porphyrins | Found in feces (in their concentration is high then toxicity in the body increases) |
| Proto porphyrins | Found in blood and tissues |
The function of Porphyrins
1. For oxygen transportation
Heme is an important metalloporphyrin. It is a pigment and worked as an Oxygen carrier. Heme transports oxygen from the entire body.
2. Mediation in the electron transport chain
Cytochromes serve as electron carriers in the electron transport chain in both plants and animals.
3. In Photosynthesis
Chlorophyll is also a pigment and a type of metalloporphyrin. It is magnesium-containing porphyrin. It helps in Photosynthesis from absorb sunlight.
4. Provide a Storage site for oxygen molecules
In muscle tissues, myoglobin provides a storage site for oxygen molecules. It is bound to an iron atom in the center of the heme group.
5. Porphyrins in cancer therapy
Porphyrins are used in the treatment of certain cancers. This technique is called cancer phototherapy. Tumors are capable of taking up more porphyrins than normal tissues. When the tumor is exposed to an argon laser, the porphyrins get excited and produce cytotoxic effects on tumor cells.
Porphyrins synthesis
- Glycine is a major precursor for the biosynthesis of Porphyrins.
- The porphyrins consist of four molecules of the mono pyrrole derivative, which is derived from two molecules of δ-amino levulinate.
- In higher eukaryotes, glycine reacts with succinyl CoA and forms alpha-amino beta ketoadipate.
- Alpha-amino beta ketoadipate decarboxylated to δ-aminolevulinate.
- 2 δ-aminolevulinate condenses to form porphobilinogen through a series of complex enzymatic reactions.
- Four molecules of porphobilinogen form protoporphyrin.
- The metal is incorporated after the protoporphyrin has been.
- Porphyrin biosynthesis is regulated by the concentration of the heme protein. It is worked as a feedback inhibitor.
- Genetic defects in the biosynthesis of Porphyrins can lead to the accumulation of pathway intermediates, causing a variety of human diseases which are known as porphyrias.
Biosynthesis of heme
- Heme is the most important porphyrin-containing compound.
- The liver and the erythrocyte-producing cells of bone marrow are the synthesizing sites of heme synthesis.
- Biosynthesis of heme occurs in the following stages
1. Formation of δ-aminolevulinate
Glycine and succinyl CoA is starting material of Porphyrin synthesis.
Where glycine is a non-essential amino acid and succinyl CoA is intermediate in the citric acid cycle.
Glycine reacts with succinyl CoA and forms δ-aminolevulinate (ALA). This reaction catalyzed by a pyridoxal phosphate-dependent δ-aminolevulinate synthase occurs in the mitochondria.
2. Synthesis of porphobilinogen
Two molecules of δ-aminolevulinate (ALA) condense to form porphobilinogen (PBG) in the cytosol.
This reaction is catalyzed by the ALA dehydratase enzyme. This is a Zn-containing enzyme and its inhibited by heavy metals such as lead.
3. Formation of porphyrin ring
Porphyrin synthesis occurs by condensation of four molecules of porphobilinogen.
In porphyrins, four pyrrole rings are connected by methylene (–CH2) bridges.
4. The interaction of two enzymes
Uroporphyrinogen I synthase and uroporphyrinogen III synthases, result in the condensation of porphobilinogen followed by ring closure and isomerization to produce uroporphyrinogen III.
5. Conversion of uroporphyrinogen III to protoporphyrin IX
This is catalyzed by a series of reactions
- Uroporphyrinogen decarboxylase decarboxylates all four acetates (A) side chains to form methyl groups (M), to produce Coproporphyrinogen.
- Coproporphyrinogen oxidase converts (oxidative decarboxylation) two of the propionate side chains (P) to vinyl groups (V) and forms protoporphyrinogen.
- Protoporphyrinogen oxidase oxidizes methylene groups (–CH2) interconnecting pyrrole rings to methenyl groups (=CH–). This leads to the synthesis of protoporphyrin IX.
6. Synthesis of heme from protoporphyrin IX
The incorporation of ferrous iron (Fe 2+) into protoporphyrin IX is catalyzed by the enzyme ferrochelatase or heme synthetase. This enzyme can be inhibited by lead.

Porphyrias
Disorders associated with heme synthesis.
Porphyrias are a group of rare inherited or acquired metabolic disorders caused by enzyme defects in the heme production pathway. Porphyrias can be grouped according to the clinical manifestations of the disease.
- Neuropsychiatric symptoms
- Cutaneous symptoms
- Neurocutaneous symptoms
Diagnosis is made by the combination of history, physical, and laboratory findings.
Neuropsychiatric symptoms: Types
- ALA deficiency porphyria (ADP)
- Acute intermittent porphyria (AIP)
- Due to the accumulation of the early heme precursors
- Abdominal pain, nausea, constipation, hypertension, psychiatric symptoms, fever, and paraesthesia.
Cutaneous symptoms: Types
- Porphyria cutanea tarda (PCT)
- Hepatoerythropoietic porphyria (HEP)
- Erythropoietic porphyria
- Congenital erythropoietic porphyria (CEP)
- Due to the accumulation of the intermediate heme precursors.
- Photosensitivity (burning of the sun-exposed skin), blisters, excess facial hair, and hyperpigmentation.
Neurocutaneous symptoms: Types
- Hereditary coproporphyria (HCP)
- Variegate porphyria (VP)
Porphyrias are either inherited or acquired and classified into two categories according to enzyme defect in the liver and erythrocytes:-
Hepatic
Occurs in enzyme defects that lie in the liver.
Erythropoietic
Enzyme deficiency occurs in the erythrocytes.
Type of porphyria’s |
Enzyme Defects |
| Hepatic porphyria’s | |
| Acute intermittent porphyria | Uroporphyrinogen I synthase |
| Porphyria cutanea tarda | Uroporphyrinogen
Decarboxylase |
| Hereditary coproporphyria | Coproporphyrinogen
oxidase |
| Variegate porphyria | Protoporphyrinogen oxidase |
| Erythropoietic porphyria’s | |
| Congenital Erythropoietic porphyria | Uroporphyrinogen III synthase |
| Protoporphyria | Ferrochelatase |
Porphyrins test
- Porphyrins test used for determination of Porphyrin levels.
- Porphyrins are responsible for the synthesis of heme in higher eukaryotes.
- Heme is a classical example of porphyrins. It is the non-protein part of hemoglobin.
- Haemoglobin is found in blood and carries oxygen molecules which means its responsible oxygen supply from each organ of our body.
- Misbalanced Porphyrins levels caused porphyria. Porphyria is a group of diseases. It is the metabolic disorder of heme synthesis.
- Porphyrin levels elevated from urine, stool, and blood samples.
- If a high level of Porphyrins is found in blood, urine, and stool samples, which is indicated in the presence of porphyria disease. Their need to check furthermore tests to find out which type of porphyria is present in their sample.
Testing for porphyrin disorders
Testing for the precursor compounds, PBG (porphobilinogen) and ALA (δ-aminolevulinic acid) utilize ion exchange chromatography to remove interference and isolate each compound.
Tests for urinary PBG and ALA
- Watson–Schwartz method uses 4- dimethylamino benzaldehyde (Ehrlich’s reagent).
- Absorbance maximum of approximately 555nm. Current assays allow quantitative measurement of analyte concentrations.
Test for porphyrins
- Enhanced fluorescence of compounds in acidic solution.
- Chromatic separation and quantification with spectrophotometry or fluorometry.
- Molecular diagnostic techniques.
Porphyrins skin
Increased excretion of uroporphyrins (I and III) caused skin diseases. It shows photosensitivity, called cutaneous photosensitivity.
This is an important clinical marker of porphyria cutanea tarda (PCT), this is also known as cutaneous hepatic porphyria.
It is associated with liver damage caused by alcohol overconsumption.
Uroporphyrinogen decarboxylase enzyme is responsible for PCT.
The liver exhibits fluorescence e due to the high concentration of accumulated porphyrins.
The following symptoms appear in this porphyria:-
- Sensitivity to sunlight causes burning skin.
- Caused redness and swelling in the skin
- Defects in skin pigmentation (skin color changed)
- Skin becomes thin and fragile
Q&A
1. What are porphyrins?
They are a complex ring structure. It is made with 4 pyrroles. Pyrroles are units of Porphyrins that consist of 4 carbon atoms and one nitrogen atom.
2. What do porphyrins do?
Are useful for the synthesis of heme protein. Heme part of hemoglobin which is responsible for oxygen transportation.
3. What prevents simple iron porphyrins from functioning as O2 carriers?
Carbon dioxide prevents simple iron porphyrins from functioning as O2 carriers.
Written By: Richa Pachori
References
1. Biochemistry 4th edition, (U. Satyanarayana U. Chakrapani)
2. Lehninger Principles of Biochemistry, 4th edition (David L. Nelson Michael M. Cox)
3. Clinical Biochemistry (William J. Marshall & Stephen K. Bangert).
